Awaiting Salvation
 |
"The problem is that the messaging is very different than what the public is being told. We're told this is going to end the pandemic. It's not going to end the pandemic.""It's going to hopefully prevent people from getting sick, but it's not going to stop transmission.""That's not what they measured."Ed Mills, clinical trials expert, part-time McMaster University professor
"There's lots still to be said in the background of the exciting news releases.""[A vaccine that provided] sterilizing immunity -- [staving off infection itself -- would be an amazing development].""[...Even one that just afforded] protective immunity [preventing people, especially the most vulnerable, from getting sick when they contract the virus, would be a] huge step forward."Dr.Gerald Evans, head, infectious diseases division, Queen's University medical school"If [infections] were not lab confirmed with routine testing in all participants, then we would not know the impact on asymptomatic disease."Dr.Julie Bettinger, professor, vaccine-safety scientist, University of British Columbia
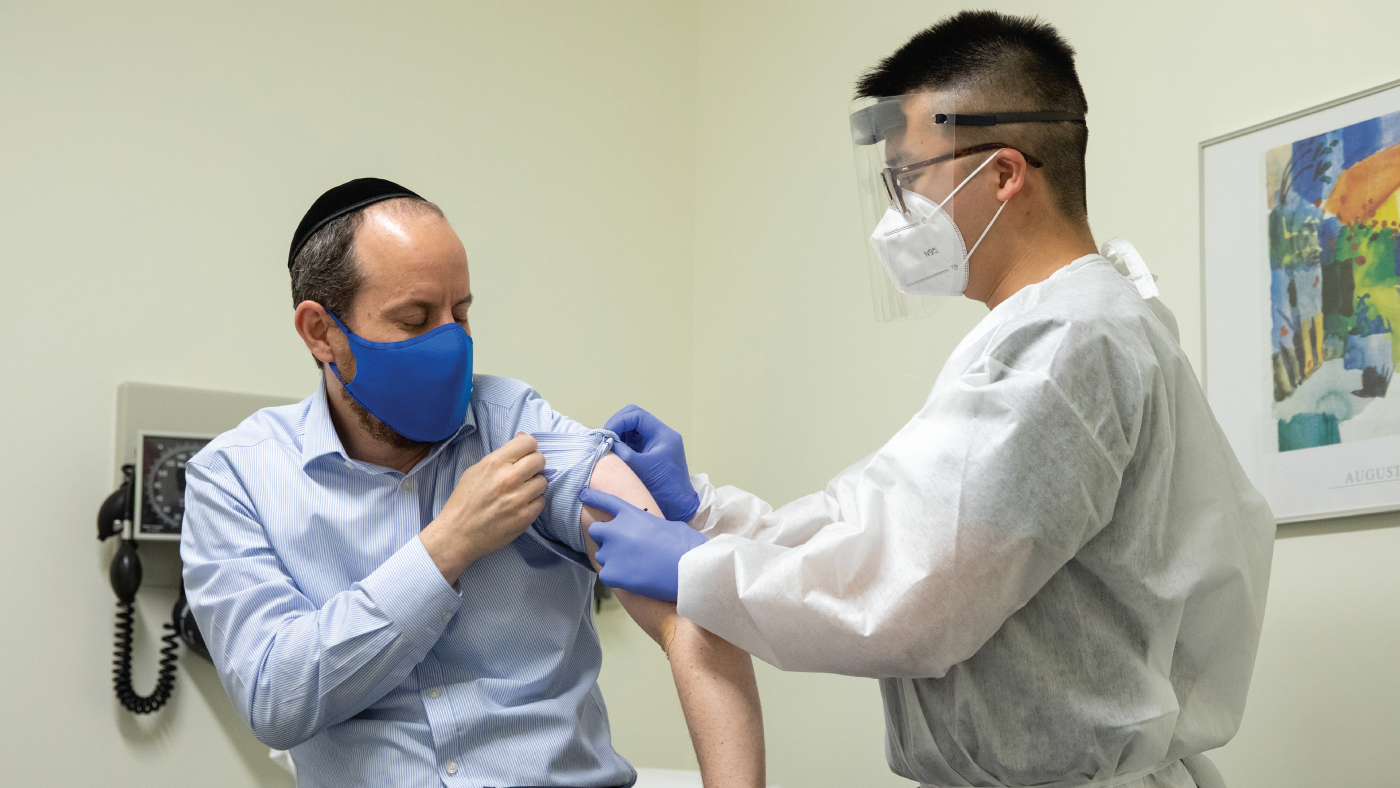 |
| Rabbi Shmuel Herzfeld has his arm disinfected by Dr. Chao Wang during a clinical trial for a coronavirus vaccine. (Amanda Andrade-Rhoades/Getty Images) |
Well then, among experts in the field of immunology some questions have arisen even while trial results reported in the past week for COVID-19 vaccines excited expectations that the global pandemic would come to an end sooner rather than later. First Pfizer, then Moderna and now AstraZeneca have proclaimed their Third-Phase Trials to have been resounding successes in an elevated sphere of reliability rare among such pharmaceutical vaccines that take much, much longer to be proven even partly successful.
On the other hand, these are self-congratulatory announcements by the very pharma-giants whose specialty is self-promotion and who stand to gain huge financial rewards from the success of their remedies to the ferociously infectious, threatening SARS-CoV-2 virus causing COVID-19 which has in the space of eight months been the cause of death of one-and-a-half-million people world-wide and stricken ill over 56 million people. Any positive news of a formula that will stop the virus is exciting.
Scientists familiar with the process of establishing the bona fides of vaccines, their efficacy and safety, are just a trifle skeptical at the same time as they are also impressed with the potential on the near horizon. Should these three companies' formulas be approved by regulators -- as they most surely will be -- the vaccines, first in line of the many being developed worldwide (an estimated 250), could establish themselves as the standard of care in the prevention of COVID-19.
Once that happens, it becomes unlikely that other laboratories will continue with their own formulas for successful vaccines; placebo trials of dozens of other products showing potential may come to a standstill, even though some of those other vaccines may have properties superior to those of the leading three first to complete their third trials. What about a vaccine that requires no refrigeration making it simpler to distribute? One that has a longer-lasting protection? One that requires one dose only?
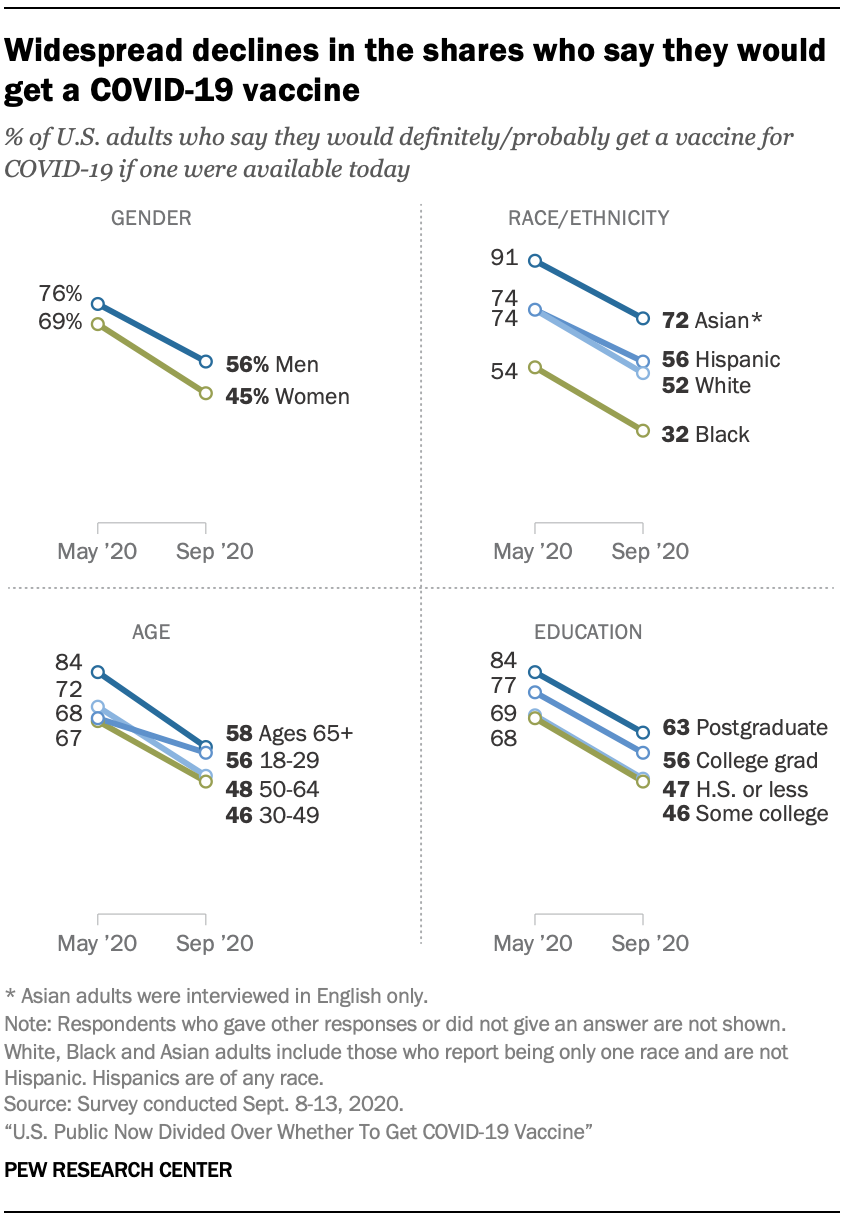 And then there is the fact that the efficacy rates boasted of are not reflective of the ability of the vaccines to prevent infection; the clinical trials of the third phase tracked cases in which participants developed symptomatic diseases. The studies highlighted that a high percentage of those contracting the virus failed to experience symptoms; even so remain capable of transmitting the disease onward.
And then there is the fact that the efficacy rates boasted of are not reflective of the ability of the vaccines to prevent infection; the clinical trials of the third phase tracked cases in which participants developed symptomatic diseases. The studies highlighted that a high percentage of those contracting the virus failed to experience symptoms; even so remain capable of transmitting the disease onward.Pfizer's and Moderna's vaccines based on messenger RNA technology injects genetic material into the body to persuade cells to produce COVID virus bits called antigens which trigger production of antibodies against the germ. Neither company has yet produced full results of their trials, nor have either published peer-reviewed papers. The possibility is, according to some experts, the vaccine prevented infection, not merely the resulting illness but results up to the present fail to demonstrate that to be the case.
Preventing infection, not just averting symptoms of severe disease or death is not a result that most vaccines accomplish, according to Dr.Scott Halperin, immunologist at Dalhousie University. Managing to 'downgrade' COVID -19 from a sometimes-deadly virus to the equivalent of a common cold would represent a resounding success by any vaccine. "To prevent infection completely is a very high bar to achieve", he comments.
And the question looms how the first vaccines' approval may affect other vaccines still in the development stages. It would represent a challenge carrying out trials comparing other vaccines to a placebo at a time when products proven effective are already widely accessible. "The vaccines in early stages of clinical trials are now as good as dead because nobody will agree to take a placebo", commented Amir Attaran, health policy expert at the University of Ottawa.
"Let's say these vaccines were out and they were available to you and somebody said to you, 'How would you like to participate in a trial where you have ... 50 percent chance of not getting a vaccine'?""How enthusiastic would you be?"Dr.Gordon Guyatt, expert in medical-research methods, McMaster University

Labels: COVID-19, Emergency Release, Infectious Diseases, Pharmaceuticals, Vaccines

0 Comments:
Post a Comment
<< Home