Brave New World of Bioscience and Application
"We are still in the early stages [of stem cell research/application]. In 2014, [scientists] at the Riken Center for Developmental Biology [Japan] had great success using iPS cells to treat macular degeneration. They took skin cells from a 70-year-old patient and derived iPS cells from them. They then differentiated the stem cells [directed them 'back down' the normal developmental path] to become adult retinal cells. These were transplanted into the patient's eye. That was a huge success."
Shinya Yamanaka, scientist, director, Kyoto University Center for iPS Cell Research and Application
 |
| Dr. Shinya Yamanaka, CiRA Director, Kyoto University |
A decade ago at Kyoto University, scientist Shinya Yamanaka pioneered a technique whereby adult cells could be "reprogrammed" back to their "pluripotent" condition at which point they had the potential to become any body cell ranging from a heart muscle cell to a neuron in the brain. This scientific process made it possible to bypass the controversy raging over the kind of research that made use of human embryos, offending the moral susceptibility of many.
And at the same time, presenting a method by which there would be no shortage of stem cells.
For this groundbreaking work, Dr. Yamanaka took his share of the 2012 Nobel Prize in Physiology or Medicine for his work in setting the stage for science to reprogram mature cells back into their former earliest stage now known as induced pluripotent stem cells [iPS cells]. Dr. Yamanaka now directs the Center for iPS Cell Research and applications at Kyoto University, Tokyo, Japan.
This internationally renowned Laureate explained in an interview some of the roadblocks encountered along the way to simply assuming that these iPS cells could be used, once successfully altered using an individual patient's cells, to be placed back into that patient's body to correct a condition related to age or chronic disease. It had been discovered through investigating the genome sequence of a patient's iPS cells that a mutation existed.
And that discovery put a stop to a process that was planned to get underway similar to the one Dr. Yamanaka spoke of in the treatment of macular degeneration for a 70-year-old patient. The realization that the pluripotent stem cells, having the capability of rapid proliferation ad infinitum posed a risk, after multiple cell cycles of mutations increasing and in the process possibly causing cancer posed a risk too great to proceed as planned.
Because of this realization Dr. Yamanaka's research laboratory addressed the issue by developing allogenic stem cell lines from donors, which would be compatible cells ready for transplantation into a patient, similar to a blood transfusion. Rigorous quality tests must be performed, with the inclusion of sequencing the stem cells' genomes to ensure the cells would be free from the presence of cancer-causing mutations.
Crudely put, stem cell treatments, originally imagined as in theory representing the kind of "personalized" medicine where the patient's own stem cells would be used in the generation of adult cells to bypass the risk of the body's immune system springing into action to reject cells other than those recognized as their own, solving an everpresent problem in transplantation. While that personalized track could still be pursued, it would be both time-consuming and hugely expensive.
 |
 |
Dr. Yamanaka also explained that the exciting prospect of stem cell therapy solving all ills had been initially rather overstated. Pointing out the limitations where roughly ten conditions only could be treated by stem cell therapy; which included Parkinson's, retinal and corneal diseases, heart and liver failure, diabetes and spinal cord injury, joint disorders and some blood disorders.
Even limited to those conditions, however, it is more than obvious that much good could come of its use, since those ten encompass quite a panoply of body malfunctions hugely impacting quality of life and for most, leading to early death.
And the reason ascribed to stem cell application limits seems simple enough, that those diseases are typically caused by loss of function of one type of cell, whereas other diseases are the result of multiple types of cell failures and thus cannot be treated with stem cell therapy. Direct cellular reprogramming describes a new strategy reprogramming adult cells back to a state specific to the organ the cells were derived from.
If, for example all the cartilage in an elderly patient's knee required replacement that is the kind of process that may improve prospects over iPSC [induced pluripotent stem cells], even while iPS would represent the first choice when treating a younger patient with only a small lesion where good cartilage could be produced from iPSCs then transplanted the purified cartilage to that small lesion.
This is most definitely an evolving, time-consuming and costly process to be fully understood and reliable before it becomes mainstream.
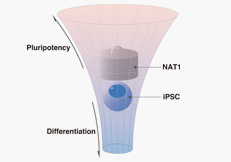
- Induced pluripotent stem cells (iPSCs) are a new type of pluripotent stem cells first generated in 2006 in mouse. They represent a potentially important resource for applications in regenerative medicine;
- PS cells generated from patient cells can be used to create cells that replicate the tissue affected by disease. This technique offers the prospect of using iPS cells to test drug efficacy, side effects, and toxicity and to develop new drugs and therapies.
Labels: Bioscience, Health, Medicine, Stem Cell Therapy

0 Comments:
Post a Comment
<< Home